Rationale
The American Heart Association (AHA) guideline recommends to increase potassium intake to reduce the risk of hypertension and cardiovascular disease.1 Because most Americans have an inadequate daily potassium intake,2 potassium supplementation is an effective strategy to increase potassium intake to control hypertension and reduce cardiovascular morbidity and mortality. Potassium chloride (KCL) supplements are frequently used to increase serum potassium levels.3
Potassium-sparing diuretics (KSD) are another approach to increase serum potassium levels through antagonism of aldosterone and include spironolactone, eplerenone, amiloride, and triamterene.4 Spironolactone or eplerenone are effective add-on therapies for a patient with poorly controlled hypertension with hypokalemia.5 Spironolactone and eplerenone are also commonly prescribed for patients with failure because of a significant reduction in cardiovascular death and sudden cardiac death report in clinical trials.6-8 Also, amiloride or triamterene are frequently prescribed as a fixed‐dose combination with hydrochlorothiazide for essential hypertension because they tend to be relatively ineffective when used as monotherapy.9
Hyperkalemia (serum potassium ≥5.5 mmol/L) is a common electrolyte abnormality and is frequently asymptomatic or non-specific mild symptoms, but severe hyperkalemia could lead to cardiac arrhythmias and death.4,10-12 Depending on the threshold used for hyperkalemia, commonly set at values than range from 5.0 to 6.0 mmol/L, incidence of hyperkalemia varies from 1% to 10%.13 In hospitalized patients hyperkalemia occurs in 1.3% to 10%, and has been associated with a mortality rate of 1 per 1,000 patients.12 Parenteral administration of potassium is a common cause of hyperkalemia in hospitalized patients.14
Drug-induced hyperkalemia is also common in everyday clinical practice.12 Both potassium chloride (KCL) and KSDs are known to increase the risk of hyperkalemia,4,10,12 and their pharmacodynamic effects are additive.13 Despite the risk of hyperkalemia, the dual use of potassium-sparing diuretic and potassium is used in clinical practice. A study of routine clinical practice found that 40% of patients with heart failure who treated with spironolactone also continue on potassium supplements despite the absence of hypokalemia.15 Therefore, the clinician should assess risk and take action if necessary if the combined use is unavoidable.
Algorithm
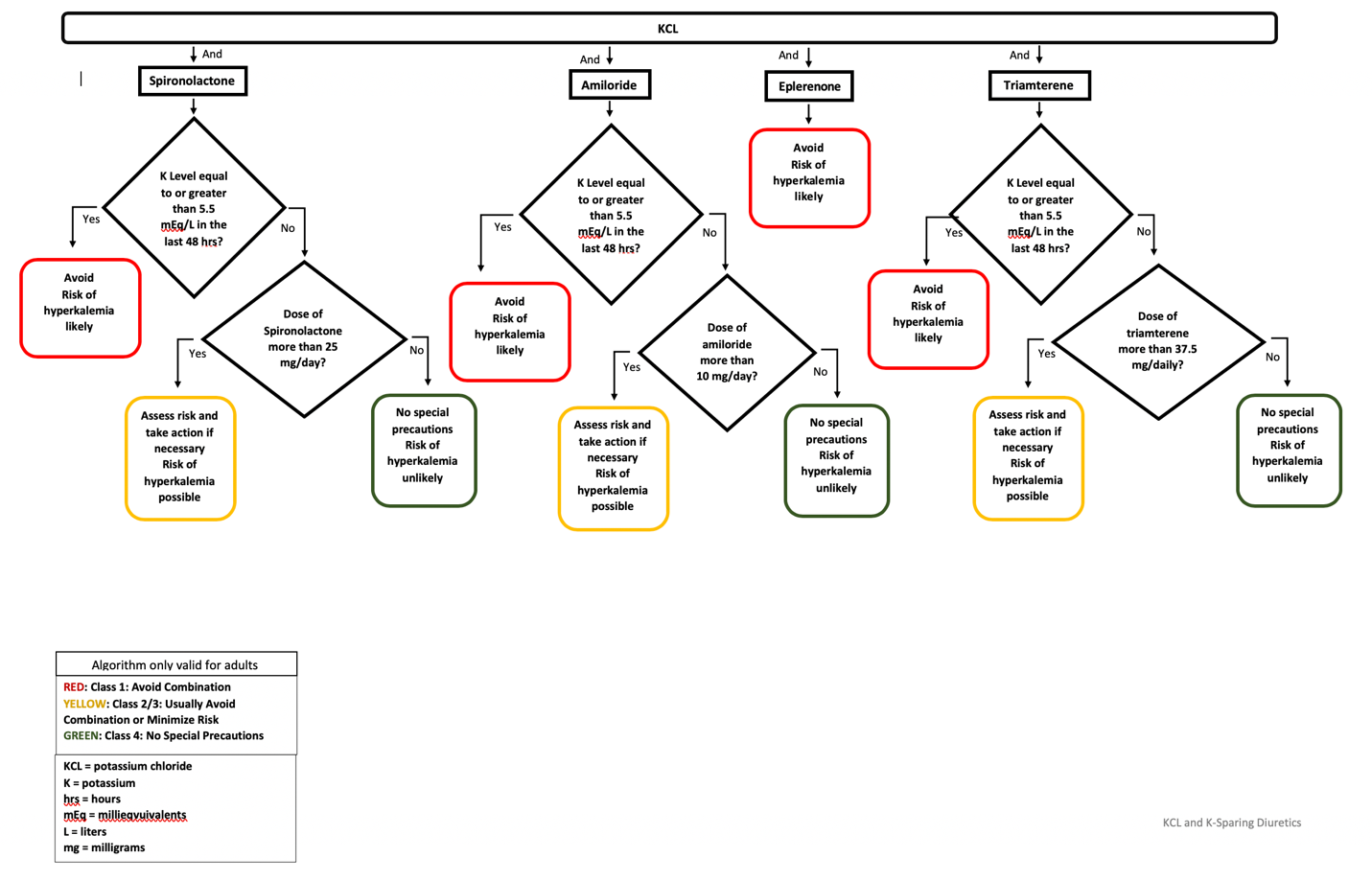
Explanation:
Although potassium supplementation, has cardiovascular benefits, it can also increase the risk of hyperkalemia. The Boston Collaborative Drug Surveillance Program demonstrated an incidence of hyperkalemia of 3.6% among 4,921 patients taking physician-prescribed potassium supplements.16 High-potassium diet, a potassium supplement, or salt substituted was also reported to be associated with hyperkalemia.5 In addition, KSDs such as spironolactone and eplerenone can cause hyperkalemia by blocking renal aldosterone receptors.12Amiloride and triamterene may cause hyperkalemia by blocking sodium reabsorption in the collecting tubule and collecting ducts which lead to a reduction in renal excretion of potassium.9,12
A retrospective study of 15,000 hospitalization patients found that KSD were strongly predictive of high serum potassium (K>5.0 mmol/L) with an OR= 1.9 (95% CI, 1.4 – 2.7) per one define daily dose (DDD).17 Another nested case-control study of 19,194 patients with new heart failure diagnoses were followed up over an average of 3.9 years (SD± 3.21 years) reported 2176 episodes of hyperkalemia. This study found the use of potassium‐sparing diuretics (including spironolactone, amiloride, eplerenone, triamterene) increased the risk of hyperkalemia by 3-fold (OR= 3.01, 95% CI 2.61–3.48) and the risk was slightly higher for spironolactone (OR= 3.23, 95% CI, 2.77–3.77) and lower for amiloride (OR= 2.63; 95% CI, 1.96–3.54).10 Moreover, the risk of hyperkalemia was the greatest during the first month of treatment (< 30 days) with an OR=9.16 (95% CI 5.99–14.02) for all potassium‐sparing diuretics, and OR=10.68 (95% CI, 6.61–17.26) for spironolactone.10 In addition, hyperkalemia seems to be dose-dependence of K-sparing diuretics. High dose of K-sparing diuretics (i.e, spironolactone > 25 mg/day, eplerenone > 50 mg/day) was reported to be associated with hyperkalemia.5 In the Randomized Aldactone Evaluation Study [RALES], patients with symptomatic heart failure who received conventional therapy of stable dose of an angiotensin-converting enzyme (ACEi), loop diuretic, and optional digitalis, the addition of 12.5-, 25-, 50- and 75-mg spironolactone increased the incidence of hyperkalemia (serum potassium ≥ 5.5 mmol/L) from 5%, 13%, 20%, and 24%, respectively.18 Another randomized control trial in patients with hemodialysis also reported more instances of hyperkalemia (0.89 events per patient-year) with 50-mg spironolactone vs. 0.23 events per patient-year with 25-mg.19 However, the mean daily dose of spironolactone was higher (40.7 ± 3.1 mg) in clinical practice compared to the RALES trial, thus, incidence of hyperkalemia was also higher (24% for serum potassium ≥ 5.5 mmol/L, 12% for serum potassium ≥ 6.0 mmol/L).15 Another case-control study in patients with congestive heart failure (CHF) hospitalization suggested that average 25-mg/day spironolactone was independently associated with hyperkalemia (K ≥ 5.6 mmol/L) with an OR= 4.18 (95% CI, 1.27–13.79).20 EMPHASIS-HF trail reported incidence of serum K >5.5 mmol/L and K > 6.0 mmol/L to be 11.8% and 2.5%, respectively in patients with heart failure receiving up to 50 mg daily of
eplerenone.21 EPHESUS trial also found a higher incidence of serious hyperkalemia with eplerenone (25 mg per day initially, titrated to a maximum of 50 mg daily) compared to placebo (5.5% vs. 3.9%; p = 0.002).8 Moreover, 50-, 100- and 400-mg/day of eplerenone can increase potassium up to 0.11, 0.08, and 0.36 mmol/L, respectively.22 Moreover, the use of eplerenone up to 50 mg daily for one month period increased potassium level 0.16±0.51 mmol/L.21 Hyperkalemia was also reported with amiloride, especially in patients with chronic kidney disease or people receiving other drugs associated with hyperkalemia including ACEI, angiotensin II receptor blockers (ARBs), and nonsteroidal anti-inflammatory drugs (NSAIDs).9 A median daily increase in serum potassium was 0.52 vs 0.40 mmol/L/day, p= 0.007 for high-dose potassium-sparing diuretics (amiloride > 10 mg/day or spironolactone > 25 mg) vs low-dose.13 Hyperkalemia also has been reported with concurrent use of triamterene and chlorothiazide or hydrochlorothiazide in congestive heart failure patients.23
Similarly, hyperkalemia seems to be dose-dependence of potassium supplement as well. A retrospective study randomly selected 600 patients with hyperkalemia (serum K≥ 5.0 mmol/L) and found that during their hospitalization the reported a mean daily increase in serum potassium was 0.48 mmol/L/day vs 0.40 mmol/L/day for high-dose potassium chloride supplements (> 3000 mg/day) vs. low dose (p=0.006).13 A multivariate analysis identified risk factors associated with the high mean daily increase in serum potassium in the following order potassium supplements (OR=3.39, 95% CI, 2.25-5.09) > severe renal impairment (OR=3.12, 95% CI, 1.74-4.01) > use of ACE inhibitors or ARBs (OR=2.64, 95% CI, 1.74-4.001) > use of potassium-sparing diuretics (OR=2.07, 95% CI, 1.31-3.25) > diabetes mellitus (OR=1.53, 95% CI, 1.01-2.31). The authors explained that potassium supplement was strongly associated with high mean daily increase in serum potassium possibly due to almost 30% of the study patients experienced hypokalemia and received aggressive treatment with potassium supplement.13 The risk of hyperkalemia for combination use of potassium chloride (KCL) and K-sparing diuretic has been suggested to be additive.13 A study analyzed prescriptions for 50,835 patients admitted to the University of Zurich between 12/1/2009 and 7/31/2011 reported that hyperkalemia occurred after a median of 3.3 days of coadministration of potassium-sparing
diuretics and potassium supplement, and incidence of hyperkalemia occurred after a median observation time of 4.5 days. The risk increased over time during prolonged coadministration.24,25
Several other factors have also been associated with the risk of hyperkalemia. These include impaired renal function,11,13,17,20,26 diabetes mellitus,5,11,13,17,20,26 infrequent serum potassium monitoring,4,11 baseline potassium level,11 angiotensin-converting enzyme inhibitors (ACEi),4,10,11,13,20,25 angiotensin receptor blockers (ARBs).11,13,25 The risk of hyperkalemia increased dramatically with severe renal impairment. In patients newly diagnosed with heart failure, compared to subjects with normal renal function, the risk of hyperkalemia increased to approximately 2-fold (OR=1.82, 95% CI, 1.53–2.16), 5-fold (OR=4.57, 95% CI, 3.82–5.46), and 16-fold (OR=16.32, 95% CI, 12.96–20.56) for eGFR values of 45–59, 30–44, and <30 mL/min/1.73m2, respectively.10 Also, diabetes mellites increases the risk of hyperkalemia approximately 2-fold (OR=1.52; 95% CI, 1.31-1.75).10 Serum K > 4.3 mEq/L was found to be an independent baseline predictor of increased serum K > 6 mEq/L.26 The risk of hyperkalemia is 3-fold higher (RR=2.93, 95% CI, 1.97-4.36) in patients with K>4.5 mEq/L compared to those with normokalaemia.25
Drug interaction algorithm
Click to download a PDF of the flow diagram

References:
1. Sacks FM, Svetkey LP, Vollmer WM, et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N Engl J Med. Jan 4 2001;344(1):3-10. doi:10.1056/nejm200101043440101
2. Aaron KJ, Sanders PW. Role of dietary salt and potassium intake in cardiovascular health and disease: a review of the evidence. Mayo Clin Proc. Sep 2013;88(9):987-95. doi:10.1016/j.mayocp.2013.06.005
3. Filippini T, Violi F, D’Amico R, Vinceti M. The effect of potassium supplementation on blood pressure in hypertensive subjects: A systematic review and meta-analysis. Int J Cardiol. Mar 1 2017;230:127-135. doi:10.1016/j.ijcard.2016.12.048
4. Chang AR, Sang Y, Leddy J, et al. Antihypertensive Medications and the Prevalence of Hyperkalemia in a Large Health System. Hypertension. Jun 2016;67(6):1181-8. doi:10.1161/hypertensionaha.116.07363
5. Sica DA. Mineralocorticoid Receptor Antagonists for Treatment of Hypertension and Heart Failure. Methodist Debakey Cardiovasc J. Oct-Dec 2015;11(4):235-9. doi:10.14797/mdcj-11-4-235
6. Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med. Sep 2 1999;341(10):709-17. doi:10.1056/nejm199909023411001
7. Struthers A, Krum H, Williams GH. A comparison of the aldosterone-blocking agents eplerenone and spironolactone. Clin Cardiol. Apr 2008;31(4):153-8. doi:10.1002/clc.20324
8. Pitt B, Remme W, Zannad F, et al. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med. Apr 3 2003;348(14):1309-21. doi:10.1056/NEJMoa030207
9. Epstein M, Calhoun DA. Aldosterone blockers (mineralocorticoid receptor antagonism) and potassium-sparing diuretics. J Clin Hypertens (Greenwich). Sep 2011;13(9):644-8. doi:10.1111/j.1751-7176.2011.00511.x
10. Michel A, Martin-Perez M, Ruigomez A, Garcia Rodriguez LA. Risk factors for hyperkalemia in a cohort of patients with newly diagnosed heart failure: a nested case-control study in UK general practice. Eur J Heart Fail. Feb 2015;17(2):205-13. doi:10.1002/ejhf.226
11. Berkova M, Berka Z, Topinkova E. Arrhythmias and ECG changes in life threatening hyperkalemia in older patients treated by potassium sparing drugs. Biomedical papers. 2014;158(1):084-091. doi:10.5507/bp.2012.087
12. Ben Salem C, Badreddine A, Fathallah N, Slim R, Hmouda H. Drug-induced hyperkalemia. Drug Saf. Sep 2014;37(9):677-92. doi:10.1007/s40264-014-0196-1
13. Indermitte J, Burkolter S, Drewe J, Krahenbuhl S, Hersberger KE. Risk factors associated with a high velocity of the development of hyperkalaemia in hospitalized patients. Drug Saf. 2007;30(1):71-80. doi:10.2165/00002018-200730010-00007
14. Perazella MA. Drug-induced hyperkalemia: old culprits and new offenders. Am J Med. Sep 2000;109(4):307-14. doi:10.1016/s0002-9343(00)00496-4
15. Bozkurt B, Agoston I, Knowlton AA. Complications of inappropriate use of spironolactone in heart failure: when an old medicine spirals out of new guidelines. J Am Coll Cardiol. Jan 15 2003;41(2):211-4. doi:10.1016/s0735-1097(02)02694-3
16. Lawson DH. Adverse reactions to potassium chloride. Q J Med. Jul 1974;43(171):433-40.
17. Henz S, Maeder MT, Huber S, Schmid M, Loher M, Fehr T. Influence of drugs and comorbidity on serum potassium in 15 000 consecutive hospital admissions. Nephrol Dial Transplant. Dec 2008;23(12):3939-45. doi:10.1093/ndt/gfn380
18. Effectiveness of spironolactone added to an angiotensin-converting enzyme inhibitor and a loop diuretic for severe chronic congestive heart failure (the Randomized Aldactone Evaluation Study [RALES]). Am J Cardiol. Oct 15 1996;78(8):902-7. doi:10.1016/s0002-9149(96)00465-1
19. Charytan DM, Himmelfarb J, Ikizler TA, et al. Safety and cardiovascular efficacy of spironolactone in dialysis-dependent ESRD (SPin-D): a randomized, placebo-controlled, multiple dosage trial. Kidney Int. Apr 2019;95(4):973-982. doi:10.1016/j.kint.2018.08.034
20. Ramadan FH, Masoodi N, El-Solh AA. Clinical factors associated with hyperkalemia in patients with congestive heart failure. J Clin Pharm Ther. Jun 2005;30(3):233-9. doi:10.1111/j.1365-2710.2005.00638.x
21. Zannad F, McMurray JJ, Krum H, et al. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med. Jan 6 2011;364(1):11-21. doi:10.1056/NEJMoa1009492
22. Weinberger MH, Roniker B, Krause SL, Weiss RJ. Eplerenone, a selective aldosterone blocker, in mild-to-moderate hypertension. Am J Hypertens. Aug 2002;15(8):709-16. doi:10.1016/s0895-7061(02)02957-6
23. Cohen AB. Hyperkalemic effects of triamterene. Ann Intern Med. Sep 1966;65(3):521-7. doi:10.7326/0003-4819-65-3-521
24. Eschmann E, Beeler PE, Kaplan V, Schneemann M, Zund G, Blaser J. Clinical decision support for monitoring drug-drug-interactions and potassium-increasing drug combinations: need for specific alerts. Stud Health Technol Inform. 2012;180:1200-2.
25. Eschmann E, Beeler PE, Kaplan V, Schneemann M, Zund G, Blaser J. Patient- and physician-related risk factors for hyperkalaemia in potassium-increasing drug-drug interactions. Eur J Clin Pharmacol. Feb 2014;70(2):215-23. doi:10.1007/s00228-013-1597-2
26. Pitt B, Bakris G, Ruilope LM, DiCarlo L, Mukherjee R. Serum potassium and clinical outcomes in the Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study (EPHESUS). Circulation. Oct 14 2008;118(16):1643-50. doi:10.1161/circulationaha.108.778811